The structural basis of Facioscapulohumeral muscular dystrophy(FSHD).
FSHD is a disabling muscular dystrophy condition caused by the ectopic expression of the toxic DUX4 protein in muscle cells. We
are studying the interactions of DUX4 with other protein partners, to understand the earliest steps in its toxicity. Our mass
spectrometry analyses (a) have revealed that DUX4 is recruiting not only the known p300 activator, but also the Mediator complex
through interaction with two of its subunits (Med25 and Med15). We are now trying to solve the atomic structures of these interactions (b)
by using XL-MS and cryo-electron microscopy. These structures may suggest a potential therapeutic intervention strategy to stop FSHD.

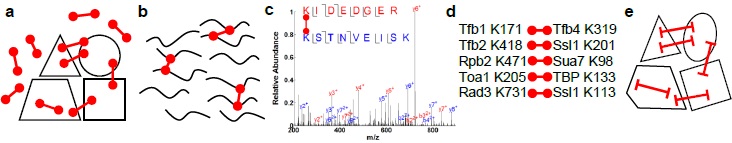
How does cross-linking and mass-spectrometry (XL-MS) work?
Cross-linking and mass-spectrometry is a powerful approach to obtain structural information
on protein complexes that are hard to resolve by cryo-Electron Microscopy or X-ray crystallography. The idea is to cross-link
the intact complex under native conditions with a short bi-functional cross-linking reagent (a). After the covalent
cross-linking occurred, the complex is denatured and digested with a protease (b). The digest is analyzed by mass-spectrometry (c),
and pairs of residues that underwent cross-linking are identified (d). Such analysis can identify hundreds of cross-linked
sites on a large complex. The cross-links are converted to distance constraints and drive computational modeling (e).